TUNNELING NANOTUBES: AN EVOLVING CONCEPT FOR CELL COMMUNICATION
SAYALI CHANDEKAR
SAVITRIBAI PHULE PUNE UNIVERSITY
chandekarsayali45@gmail.com
Introduction to Tunneling Nanotubes (TNTs):
Cell to cell communication is one of the well-known hypotheses in the field of biotechnology, where one cell communicates with another through various signals or synapses. A heterogeneous intracellular connection identified as “Tunnelling Nanotubes” (TNTs) represented a novel route of cell-to-cell communication. It was first witnessed by Rustom and his colleagues (Giulia Pinto, 2020) in rat kidney cell culture (pheochromocytoma PC12 cells).
TNTs or membrane nanotubes are described by their long, thin membranous morphology. TNTs were found to be connecting distant cells, enabling the transfer of cellular vesicles, proteins, mitochondria, miRNAs, and viral particles (HIV, herpesvirus). Also, they were found in a variety of cell types such as immune cells, neurons, endothelial cells, and mesenchymal stromal cells (MSCs).
To define TNTs, three phenotypic criteria were proposed: (i) they connect at least two cells, (ii) they contain F-actin, and (iii) they do not touch the substrate. These criteria distinguish TNTs from F-actin-rich structures such as filopodia. Electron microscopy revealed the thickness and the diameter of TNTs, which ranged from 50-200nm in PC12 cells and 180-380nm in T-cells. A study performed by Devis and his group (Maeva Dupont, 2018) learned that macrophages showed a unique way in the formation of TNTs, as thin TNT (<0.7 μm in diameter), containing only F-actin; and thick TNTs (>0.7 μm), containing F-actin and microtubules.
TNTs were also found to be involved in the key functioning of immune cells, such as activating T cells, production of various cytokines and antibodies, and increasing phagocytic activity. Therefore, TNTs can be considered an important therapeutic target in immunotherapy. However, the difference in cytoskeleton composition of TNT indicates the existence of different mechanisms of TNT formation and also provides its functional diversity.
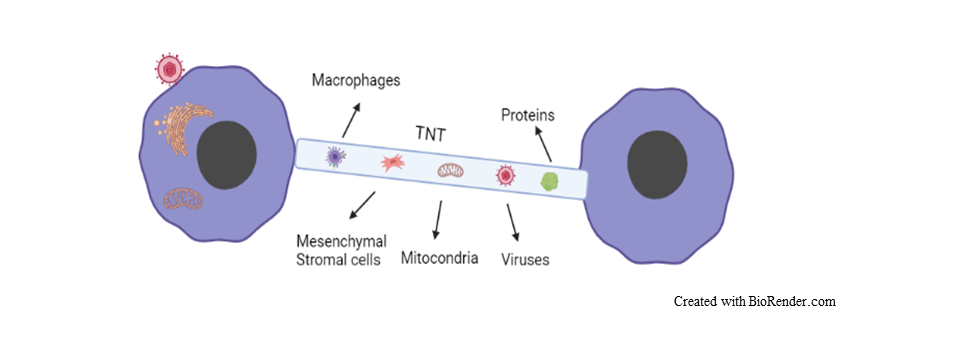 Figure 1: Cell-to-cell communication and transfer of various substances via TNT.
Figure 1: Cell-to-cell communication and transfer of various substances via TNT.
Mechanism of formation of TNTs:
There are possibly two mechanisms that support TNT formation:
- Protrusion extension mechanism: A single cell with a bulged thread is present and extends until reaching a neighbouring cell, forming TNT and making a contact.
- Cell dislodgement mechanism: two cells are initially connected through a thin thread of membrane, which elongates on cell separation to form TNT. Whereas, to form these extensions TNT first undergoes the formation of closed-ended TNT and open-ended TNT which are described as Fig 2.
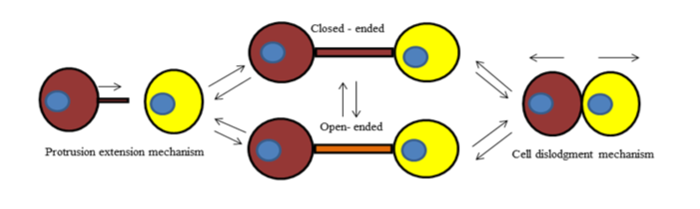
Figure 2: The two processes (1) Protrusion extension mechanism (2) Cell dislodgement mechanism showing the formation of tunneling nanotubes.
- Closed-ended TNT can form a junctional border with the target cell. These can be visualized scanning electron microscopy
- The cytoplasm of the two connected cells can be mixed which represents open-ended TNT and on the other hand to identify an open-ended TNT lipophilic dye (DiO) is used.
Formation of TNT can also take place in stress conditions such as cellular stress, oxidative stress, intracellular high calcium concentration, hypoxia, and release of excess reactive oxygen species (ROS). Researchers also identified various proteins which participated in TNT formation such as M-sec/ERp59,p53/Akt/mTOR, myosine10. The absence of these proteins showed no signs of TNT formation. .In short, the formation of TNT is not only regulated by cellular stress conditions but also due to mechanical force exerted by cells.
Conclusion:
To summarize, there has been significant progress in understanding the formation and mechanism of TNT. As the role of TNTs is described in various aspects of biology it can be further used as a potential therapeutic target in the field of immunotherapy, tissue repair, and virus-mediated diseases.
Reference (Jun-21-A6)
Author Biography
Aim: To explore the field of Biotechnology as it is the future of growing world.
Education: Post Graduate student in Biotechnology and now perusing PhD in Biotechnology from Institute of Bioinformatics and Biotechnology, Savitribai Phule Pune University
Projects: Working on Bone Biology

