RISE OF ZOMBIE GENE IN ELEPHANTS
ISHITA TARNEKAR
THADOMAL SHAHANI ENGINEERING COLLEGE
tarnekarishita@gmail.com
Cancer risk is a numbers game. It is assumed that the risk of developing cancer increases with an increasing number of cells and lifespan of an organism. The larger the body, the more the cell divisions and the more the chances of developing cancer, right? Some studies also show that large dog breeds have higher rates of cancer than smaller breeds, and in the case of humans, an increase in adult height increases the risk of human cancers.
But in 1977, an epidemiologist discovered that there is no correlation between body size and cancer risk across species. It was stated by Richard Peto and is commonly known as Peto’s Paradox. Large-sized elephants protect themselves from cancer by expressing some unique genes that destroy the damaged cells. This way, Elephants have provided a fascinating solution to Peto’s paradox by developing specialized cancer suppression mechanisms.
Elephants and Cancer
According to the statistics, only 4.81% of elephants in captivity die from cancer. It is strange since on average, elephants live for 70 years and roughly have 100 times more cells than humans. During cell divisions, genes replicate and sometimes express non-functional versions called pseudogenes or dead genes. The genome of an elephant contains LIF pseudogenes, specifically LIF6. The multifunctional interleukin-6 class cytokine leukemia inhibitory factor (LIF) can function either as a tumor suppressor or an oncogene. Activation of LIF6 may have contributed to the evolution of enhanced cancer resistance in the elephant lineage.
TP53 is another crucial tumor suppressor gene that encodes for the transcription factor, p53. Mutation in p53 is seen in a majority of cancers. In response to DNA damage, p53 proteins switch on LIF6, which forms LIF6 proteins. This causes mitochondrial dysfunction resulting in permeabilization and release of pro-apoptotic protein such as cytochrome c. This further leads to the collapse of Matrix metalloproteins (MMP), an enzyme that cleaves the elastin matrix thereby causing mitochondrial swelling, rupture, and cell death. However, the exact molecular mechanism by which LIF6 induces apoptosis is still unclear.
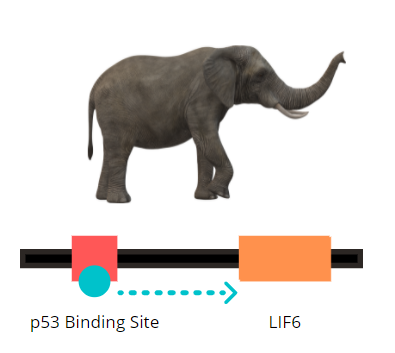
In 2015, Abegglen LM, and a team of researchers proved that the elephant genome has about 20 extra duplicates of gene TP53 (40 alleles) that trigger cell death. In contrast, other animals and humans have only one copy (2 alleles) of the same. Due to a large number of TP53 genes, multiple copies of p53 are formed. These enhance LIF6 expression thereby making cells significantly more sensitive to DNA damage. The dead gene, LIF6 came back to life and evolved gradually. Thus was termed as “zombie gene”. Beyond belief, science has witnessed the reanimation of dead genes that can produce proteins that help in cancer prevention in elephants and perhaps increase possibilities for research.
How does this concern us?
Cancer development is an evolutionary process. The cells inside the tumors keep evolving, so we have to as well. We are living amidst the Earth’s sixth mass extinction where we need mechanisms to prevent the progression of tumors and battle cancer. Correcting an error is a powerful mechanism. So maybe we can try to mimic the behavior of LIF6 expression of elephants. If p53 is found effective in mammals, we can also try to add extra copies of the same in stem cell therapies and try to treat humans. If in a particular species we discover a potential mechanism for cancer suppression, there is always a chance and broad area of scope that we can find new therapeutic targets and approaches to prevent cancer and save human lives.
Reference (Aug-21-A2)

